Research
Removal of toxic metals from wastewater using solid catalysts
In recent years, the concentration of metal ions in mining and industrial wastewater has come to be strictly regulated, and improving the efficiency of removal treatment has become an important issue. In particular, toxic metal ions such as arsenite and selenate are extremely difficult to remove by general methods such as adsorption and precipitation due to their unique properties. Therefore, oxidation treatment in water (arsenite) and reduction treatment in water (selenate) are used as pretreatment to convert them into arsenate and metallic selenium, respectively, before removal. However, these oxidation and reduction reactions are very difficult and are currently performed under very harsh conditions. Thus, improving the efficiency of the removal process of metal ions containing toxic substances has become one of the challenges in the mining and industry.
Under this background, we found that the oxidation of arsenite (As(III)) in water and the reduction of selenate (Se(VI)) in water were significantly enhanced by using a solid catalyst. The figure below shows the results of As(III) oxidation using dissolved oxygen as the oxidant. Without a catalyst, the oxidation of As(III) did not proceed with a weak oxidant such as dissolved oxygen. However, the solid catalysts, especially supported platinum catalysts, dramatically accelerated the oxidation reaction. Similarly, although not shown in the figure, supported Pt catalysts dramatically accelerated the reduction of Se(VI) in water using hydrazine as the reducing agent. These results break the scientific stereotype that solid catalysts do not contribute much to the oxidation and reduction of metal ions in water.
Although the figure below shows the reaction results in a batch reactor, we have also shown that As(III) can be fully oxidized continuously using a tube reactor packed with solid catalysts. In particular, we have demonstrated that arsenite, a common concentration discharged from copper mines, can be continuously oxidized to below the Japanese environmental standard using only dissolved oxygen in the water.
We have also been conducting research on the elucidation of the reaction mechanism. For example, it was revealed that the As(III) oxidation reaction proceeds by the Langmuir-Hinshelwood mechanism in which arsenite and oxygen are competitively adsorbed on the catalyst surface. Many interesting findings were also obtained, such as the fact that Fe(III) promoted As(III) oxidation by the oxygen over the Pt catalyst while Fe(III) alone cannot oxidize As(III). These results are expected to lead to technological developments not only in the oxidation and reduction of arsenite/selenate, but also in wastewater purification.

Catalytic conversion of hydrocarbons containing sulfur species into valuables
Fossil resources such as natural gas have sulfur species, which cause equipment corrosion and catalyst deterioration in chemical processes, so sulfur species are generally removed in advance through a desulfurization process. On the other hand, small natural gas fields are aggressively developed, and we consider that the investigation of the influence of sulfur species on various reactions is valuable in the future. We have investigated the catalytic reaction of sulfur species (hydrogen sulfide, thiols, and sulfides) and their influences on catalytic reactions. For example, sulfur species deteriorate the catalytic activity in ethylene aromatization. We have found that Ga-modified zeolite shows stable activity in spite of a trace amount of sulfur species in ethylene aromatization.
Promotion of chemical recycling of carbon fiber reinforced plastics (CFRPs)
Carbon fiber reinforced plastics (CFRPs) have excellent material properties such as light weight and high strength. The issue is that there is no effective method for CFRP recycling. To address the issue, we have investigated CFRP recycling processes in two approaches.
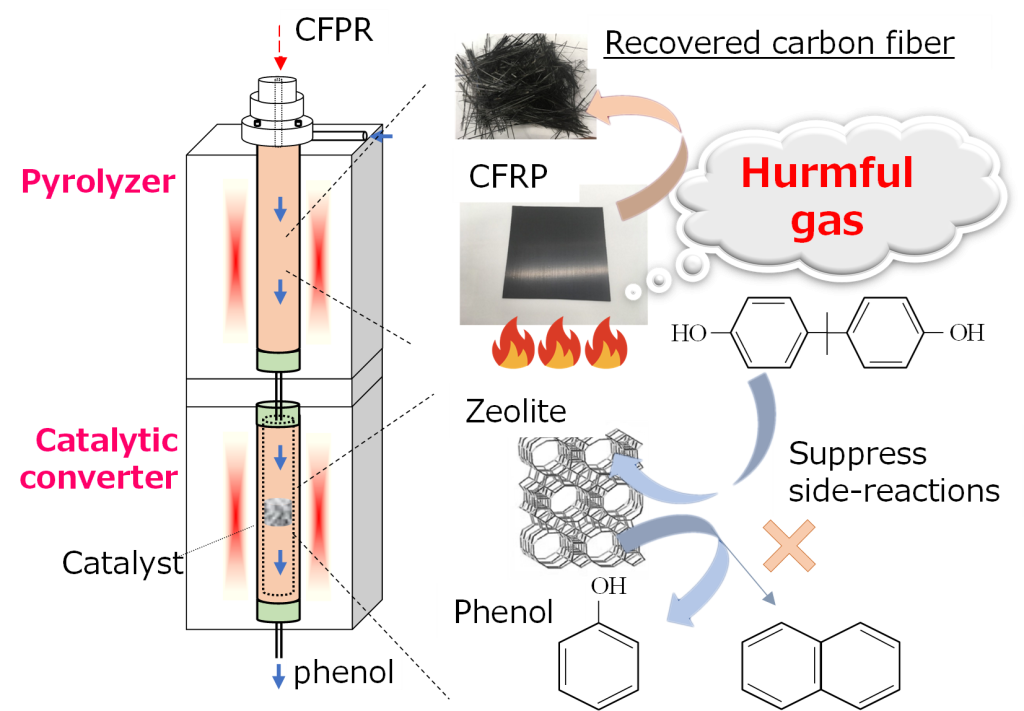
Catalytic recovery of valuables from CFRP pyrolysis gas
CFRP pyrolysis is currently the most practical recycling method. In the process, the generation of harmful gases is one of the problems because of the low temperature to avoid thermal damage to carbon fibers. We have investigated the catalytic conversion and removal of harmful gases using a zeolite catalyst. We found that high Si/Al zeolite showed high phenol selectivity, and we revealed the reaction mechanism. This research has been published in Industrial & Engineering Chemistry Research.
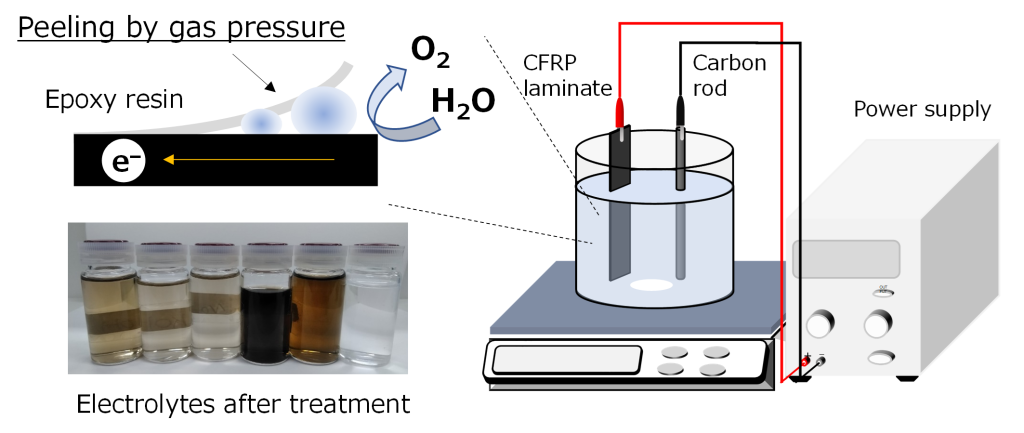
A new CFRP recycling process by an electrical treatment method
In order to develop a new recycling process that can avoid damage to carbon fibers, we have investigated a voltage application method. We found that by applying a direct current to CFRP, carbon fibers can be recovered without damage at room temperature and pressure. And we also revealed that the separation mechanism involves mechanical peeling of the resin due to the pressure of oxygen gas generated by water electrolysis. This research has been published in in Separation and Purification Technology.
Unique exothermic phenomena from complex metals in hydrogen
Recently, it has been reported that composite metals in hydrogen generate peculiar heat. A typical example of this phenomenon was discovered by Professor Emeritus Akito Takahashi of Osaka University and Professor Emeritus Akira Kitamura of Kobe University, who found that Pd-Ni-Zr alloy (PNZ alloy) samples oxidized on the surface in air exhibit sustained heat generation in hydrogen at around 300°C for several weeks.
We have also examined the exothermic behavior of a similar PNZ alloy sample in hydrogen using differential scanning calorimetry (DSC). At the same time, the hydrogen absorption behavior, alloying behavior, and amorphization behavior of the alloys were also investigated to determine their exothermic relevance. As a result, it was found that the heat generation from the PNZ sample is different from both the heat of hydrogen absorption and the heat of alloying.
We used DSC to continuously measure PNZ alloy samples in hydrogen and helium. The difference in thermal output in hydrogen and helium is shown in the figure below. The left figure shows the result of heating to 250°C and holding for 4 hours, and the right figure shows the result of heating to 400°C and holding. This PNZ sample absorbs hydrogen at around 200°C, and an exothermic peak is observed during the temperature rising process. As shown in the left figure, when the temperature was held at 250°C, where hydrogen absorption had not yet been completed, the heat output was zero and no heat was generated. On the other hand, as shown in the right figure, when the temperature was hold at 400°C, where some of the hydrogen was desorbed and heat absorption was observed, the heat output switched from endothermic to exothermic and was maintained for a long time (more than one day at other run). Furthermore, as the hydrogen pressure in the system increased, this sustained exothermic output decreased. Therefore, it was suggested that this sustained heat generation was different from the heat of hydrogen absorption.
Since the alloy phase of this sample changed little by little even at around 400°C, the change rate was also examined by XRD. However, there was no correlation between the rate of phase change and the output of the sustained heat.
As described above, the reaction mechanism of this sustained heat generation is still unknown and very interesting. We are continuing the research on this phenomenon because of its potential contribution as a new energy source.
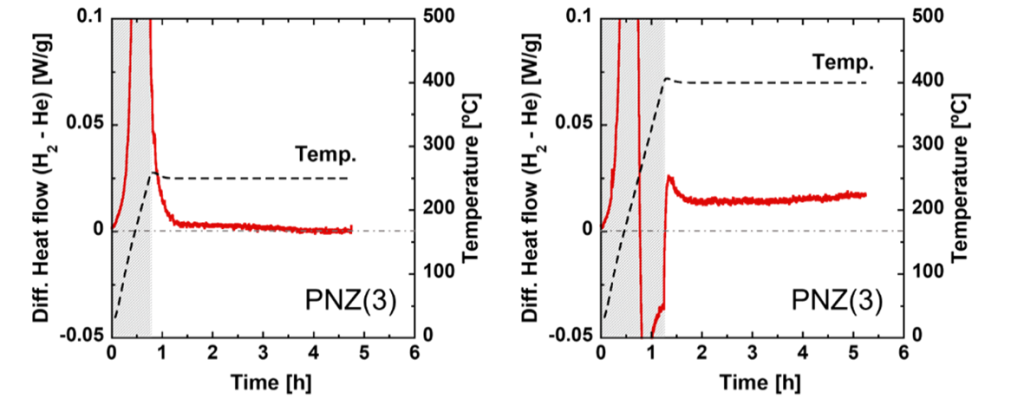
Figure Sustained heat generation from PNZ alloy at 400 ºC in hydrogen.




